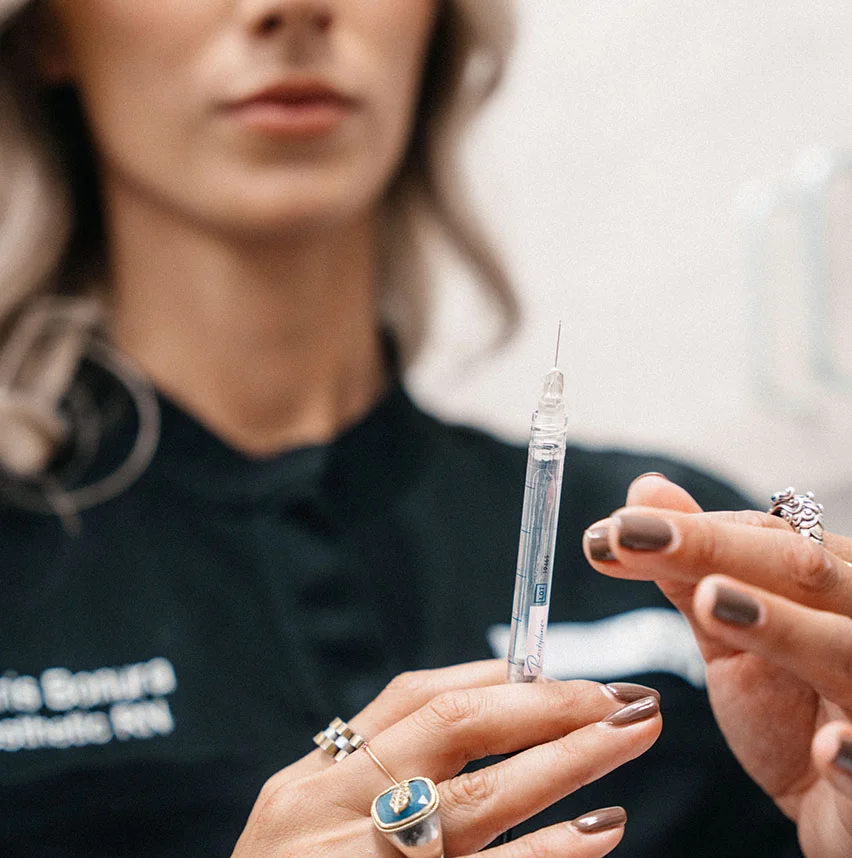
Introduction to Dermal Fillers
As we age, our collagen and elastin levels naturally decrease, which leads to wrinkles and folds. If you have been searching for a non-surgical product that can improve facial volume and smoothen away the wrinkles and age lines in your face, Dermal Fillers may be the solution you are looking for. At KH Plastic Surgery in New York City, our Dermal Fillers will fill in these areas and restore any volume loss to give our patients a smoother look. To find out if you’re a qualified candidate, please keep reading the information we have provided below.